Life Science customers with obligations under GxP requirements need to ensure that quality is part of manufacturing and controls during the design, development and deployment of their GxP-regulated product. GxP compliance for applications and infrastructure qualification and validation can often be perceived as a heavy-lift, hindering organizations who want to adapt cloud-enabled technologies to accelerate their business.
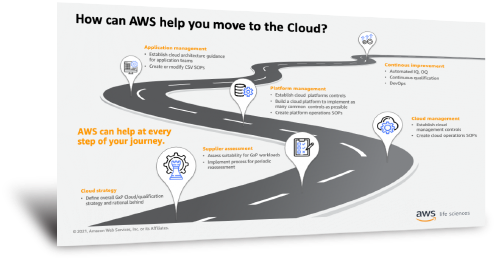
This webinar provides information on how AWS approaches GxP-related compliance and security and provides customers guidance on using AWS Services in the context of GxP. The GxP on AWS solution helps customers start their cloud journey by enabling them to onboard several AWS services which establish the environment needed to maintain compliance.
What you'll learn:
This webcast will cover the following key topics:
- GxP compliance challenges
- Why AWS for GxP compliance
- The GxP compliance journey
- A technical deep dive into how can AWS can help
- Resources you can use